Natco Pharma, one of the few Indian companies to venture into new drug discovery research, will market Bristol-Myers Squibb's chronic hepatitis C medicine-Daclatasvir Dihydrochloride (Daclatasvir) under its own brand NATDAC, and through its strategic partners in India.
The company has signed a nonexclusive, royalty free licensing agreement with the Medicines Patent Pool (MPP) and Bristol-Myers Squibb to manufacture and sell generic versions of Daclatasvir.
The company said, "This agreement allows Natco to expand access to these chronic hepatitis C medicines in 112 developing countries."
"Under the license, Natco can set its own price for the generic products it produces," it added.
Daclatasvir, discovered and developed by Bristol-Myer Squibb, is the first-in-class NS5A inhibitor used in combination with Sofosbuvir for the treatment of patients with chronic hepatitis C virus (HCV) genotype 3 infection.
Compared to other treatment options, this combination not only increases the cure rate, but is also regarded as a valuable treatment option in some of the difficult-to-treat HCV patient subsets.
 The
company has signed a nonexclusive, royalty free licensing agreement
with the Medicines Patent Pool (MPP) and Bristol-Myers Squibb to
manufacture and sell generic versions of Daclatasvir.
The
company has signed a nonexclusive, royalty free licensing agreement
with the Medicines Patent Pool (MPP) and Bristol-Myers Squibb to
manufacture and sell generic versions of Daclatasvir.
The company has signed a nonexclusive, royalty free licensing agreement with the Medicines Patent Pool (MPP) and Bristol-Myers Squibb to manufacture and sell generic versions of Daclatasvir.
The company said, "This agreement allows Natco to expand access to these chronic hepatitis C medicines in 112 developing countries."
"Under the license, Natco can set its own price for the generic products it produces," it added.
Daclatasvir, discovered and developed by Bristol-Myer Squibb, is the first-in-class NS5A inhibitor used in combination with Sofosbuvir for the treatment of patients with chronic hepatitis C virus (HCV) genotype 3 infection.
Compared to other treatment options, this combination not only increases the cure rate, but is also regarded as a valuable treatment option in some of the difficult-to-treat HCV patient subsets.
Natco Pharma,
one of the few Indian companies to venture into new drug discovery
research, will market Bristol-Myers Squibb's chronic hepatitis C
medicine-Daclatasvir Dihydrochloride (Daclatasvir) under its own brand
NATDAC, and through its strategic partners in India.
 The
company has signed a nonexclusive, royalty free licensing agreement
with the Medicines Patent Pool (MPP) and Bristol-Myers Squibb to
manufacture and sell generic versions of Daclatasvir.
The
company has signed a nonexclusive, royalty free licensing agreement
with the Medicines Patent Pool (MPP) and Bristol-Myers Squibb to
manufacture and sell generic versions of Daclatasvir.
The
company said, "This agreement allows Natco to expand access to these
chronic hepatitis C medicines in 112 developing countries."
"Under the license, Natco can set its own price for the generic products it produces," it added.
Daclatasvir,
discovered and developed by Bristol-Myer Squibb, is the first-in-class
NS5A inhibitor used in combination with Sofosbuvir for the treatment of
patients with chronic hepatitis C virus (HCV) genotype 3 infection.
Compared
to other treatment options, this combination not only increases the
cure rate, but is also regarded as a valuable treatment option in some
of the difficult-to-treat HCV patient subsets.
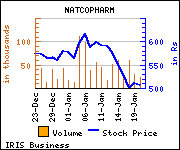
Shares
of the company gained Rs 19.1, or 3.77%, to trade at Rs 526.00. The
total volume of shares traded was 49,912 at the BSE (11.34 a.m.,
Thursday).
- See more at:
http://www.myiris.com/news/sector/natco-pharma-to-market-generic-daclatasvir-drug-in-india/20160121104200199#sthash.9gmY0CGp.dpuf
No comments:
Post a Comment